1. Modeling ion transfer across water-oil interfaces
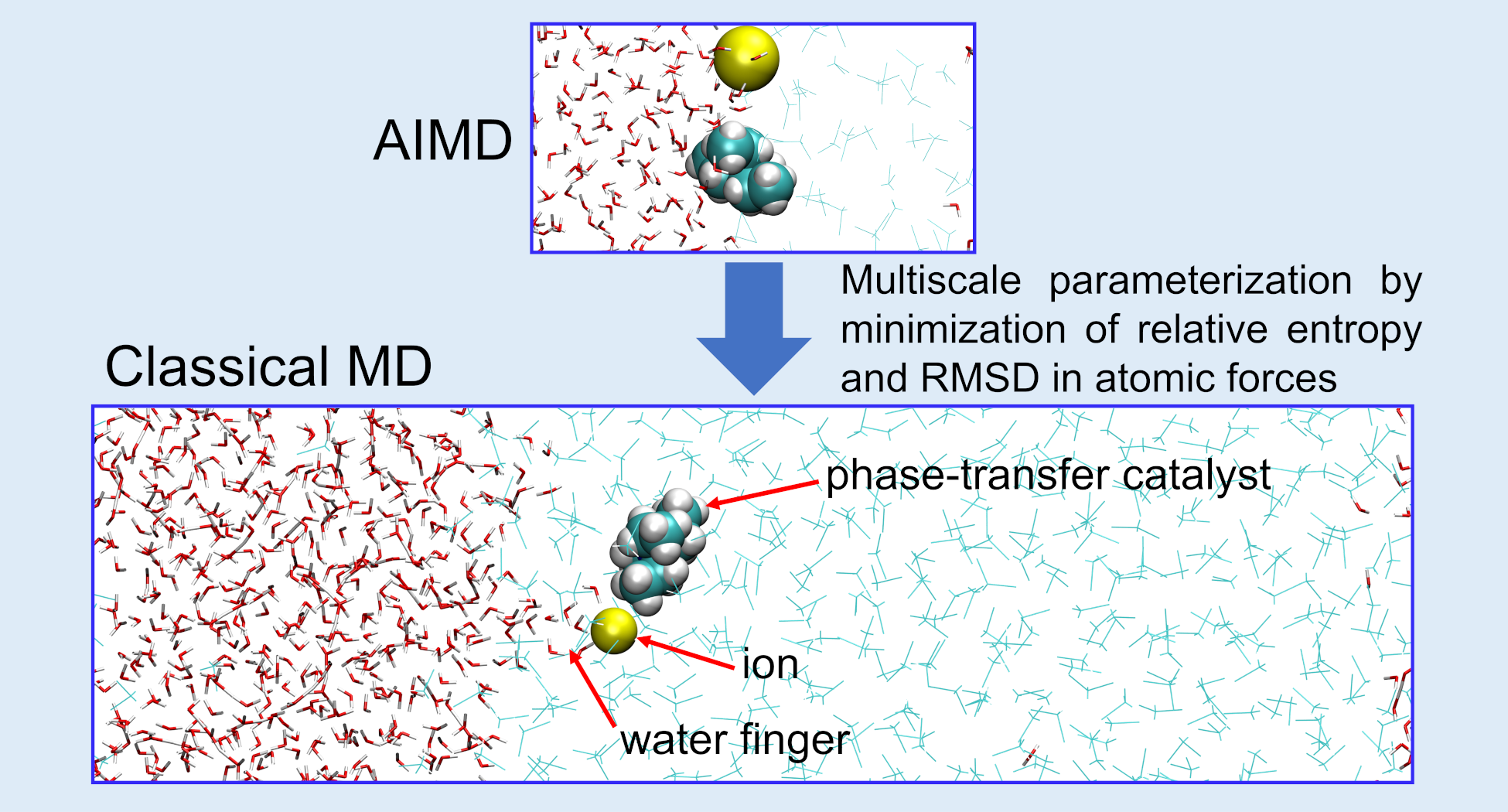
This research focuses on developing accurate and efficient molecular simulation models for liquid–liquid interfaces, especially for problems involving ion transfer, solvation, and phase-transfer catalysis. A central challenge in this area is that conventional force fields are often parameterized for bulk liquids and may not reliably describe the changing environment at an interface, where molecules experience strong variations in polarity, solvation, and electrostatic interactions. To address this, we build polarizable force fields that explicitly account for how molecular charge distributions respond to their surroundings. These models are trained using ab initio molecular dynamics reference data and optimized with statistically rigorous targets, including relative entropy and atomic force matching, so that they reproduce both structural ensembles and intermolecular forces. This multiscale strategy bridges the gap between the accuracy of first-principles simulations and the efficiency of classical molecular dynamics, allowing us to study larger interfacial systems and slower molecular processes that are otherwise inaccessible. More broadly, this research direction aims to uncover the microscopic mechanisms governing ion transport across immiscible liquid phases, including hydration-shell reorganization, interfacial fluctuations, and free-energy barriers, while also providing a general framework for constructing transferable simulation models for complex soft-matter and chemical systems.
2. Water Droplet Coalescence
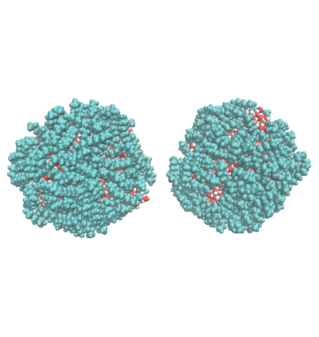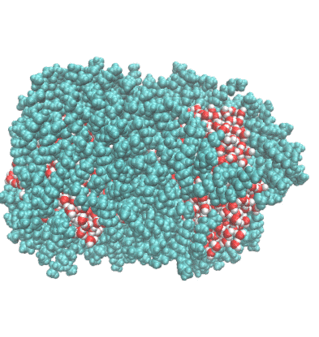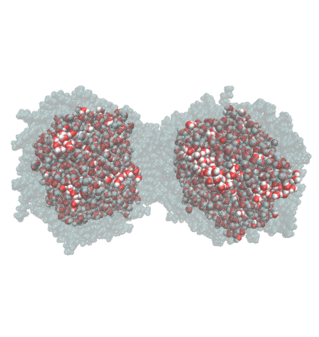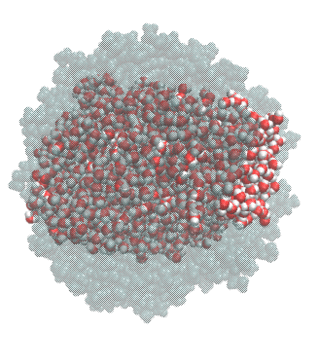
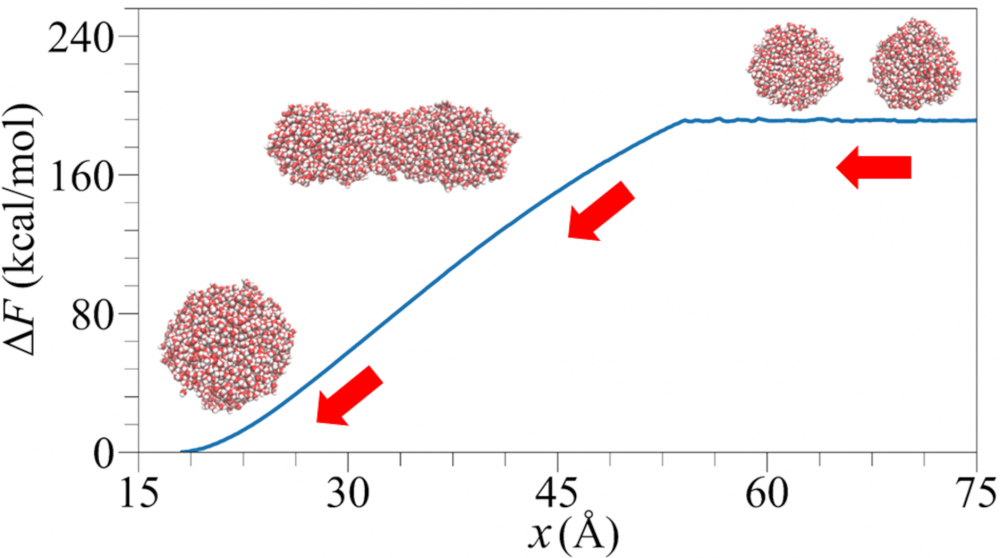
Droplet coalescence is a process in which two or more particles or droplets merge to form a larger one permanently. It has received significant attention in recent years due to its importance in cloud droplet growth and raindrop formation in atmospheric sciences. In addition to the coalescence of pure water droplets, how organic matter in the atmosphere affects coalescence is also a major concern. In this project, we are interested in understanding coalescence by examining the thermodynamics as well as the dynamics via molecular simulations.
3. Hindered diffusion near interfaces
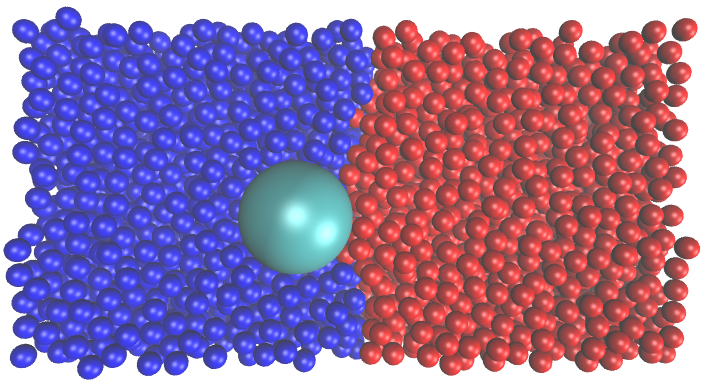
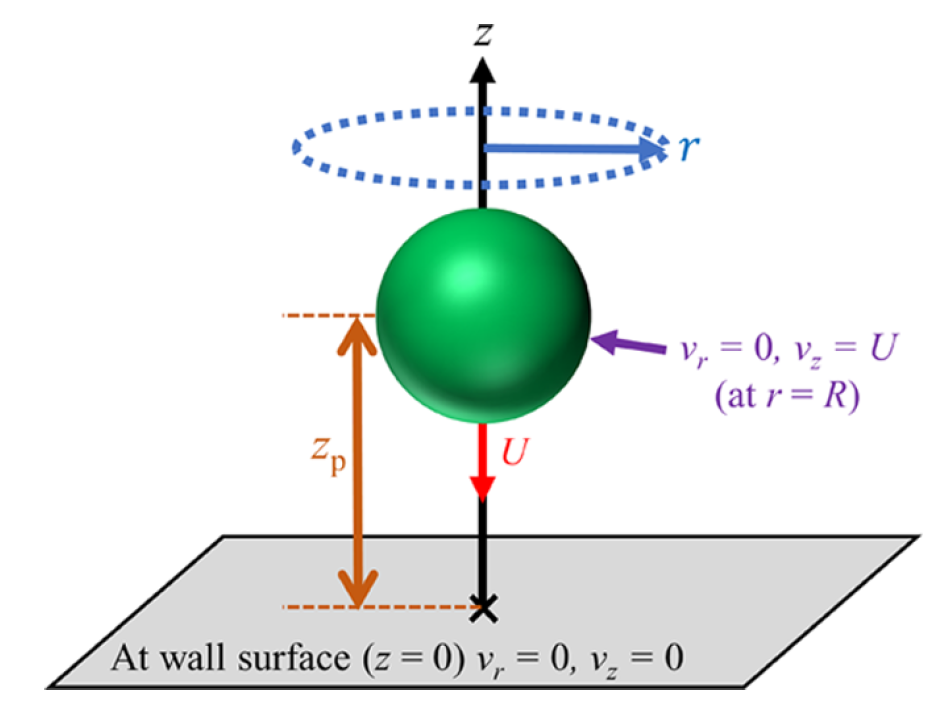
Another research direction in our group examines hindered diffusion of colloidal particles near interfaces, with the broader goal of understanding how confinement, interfacial structure, and molecular-scale interactions alter particle transport. In this line of work, we study both fluid–solid and fluid–fluid interfaces, where particle motion becomes strongly anisotropic and often slows dramatically as an interface is approached. Our simulations show that this behavior cannot always be understood from continuum hydrodynamics alone. Near fluid–solid interfaces, we found that classical hydrodynamic predictions capture the overall trend of reduced diffusivity, especially when diffusion is normalized appropriately, but important discrepancies emerge very close to the wall where molecular roughness, solvent layering, slip, and finite interaction ranges become significant. Near fluid–fluid interfaces, we observed a similarly pronounced slowdown, particularly in the direction perpendicular to the interface, and traced it to interfacial density inhomogeneity and depletion-like forces that are absent from simple continuum descriptions. Methodologically, this research combines molecular simulation, free-energy analysis, and position-dependent transport calculations to connect microscopic interfacial structure with emergent dynamical behavior.
4. Chemical kinetics of catalyzed asymmetric organic reactions (in collaboration with Prof. Yeung-Ying YEUNG)
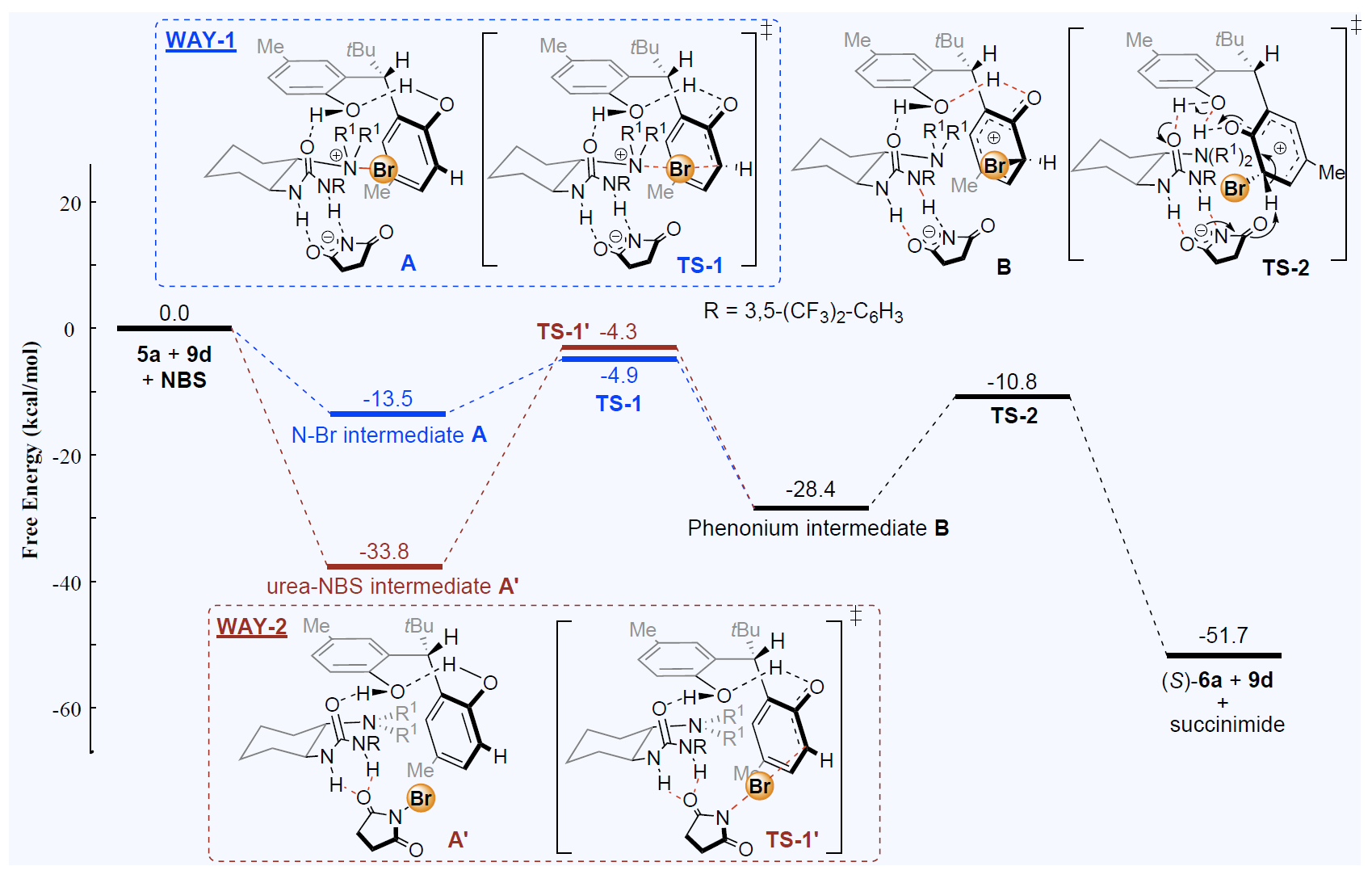
With the help of theory and computation, we can further our chemical insight into catalysts and improve them. In collaboration with Prof. Yeung-Ying YEUNG at CUHK, our group uses a variety of quantum chemistry tools including electronic structure calculations and ab initio molecular dynamics (AIMD) to understand the reaction mechanisms of how different organic catalysts carry out their functions in asymmetric synthesis, in which specific chiral compounds are made and separated. Our group is also interested in applying multi-scale modeling, using theories based on statistical mechanics, to account for explicit solvent effects.
Ying-Lung Steve Tse
Room 246 Science Centre
Office: (852) 3943 6373
stevetse@cuhk.edu.hk
